Abstract
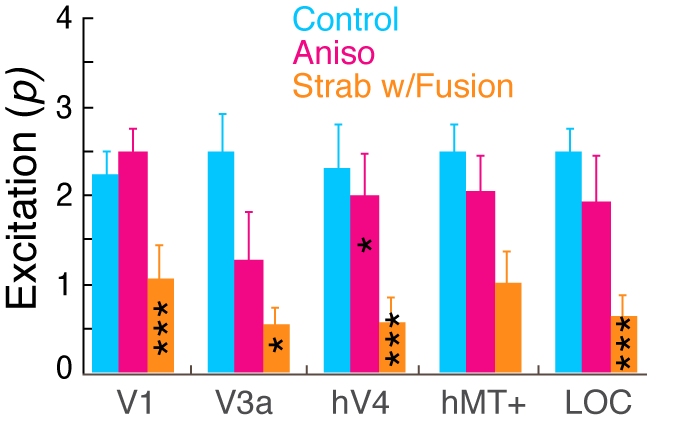
Binocular summation in strabismic amblyopia is typically reported as being absent or greatly reduced in behavioral studies and is thought to be due to a preferential loss of excitatory interactions between the eyes. Here, we studied how excitatory and suppressive interactions contribute to binocular contrast interactions along the visual cortical hierarchy of humans with strabismic and anisometropic amblyopia in both sexes, using source-imaged Steady-State Visual Evoked Potentials (SSVEP) over a wide range of relative contrast between the two eyes. Dichoptic parallel grating stimuli modulated at unique temporal frequencies in each eye allowed us to quantify spectral response components associated with monocular inputs (self-terms) and the response components due to interaction of the two eyes’ inputs (intermodulation, IM-terms). While anisometropic amblyopes revealed a similar pattern of responses to normal-vision observers, strabismic amblyopes exhibited substantially reduced IM responses across cortical regions of interest (ROIs: V1, V3a, hV4, hMT+ and lateral occipital cortex), indicating reduced interocular interactions in visual cortex. A contrast gain control model that simultaneously fits self- and IM-term responses within each cortical area revealed different patterns of binocular interactions between individuals with normal and disrupted binocularity. Our model fits show that in strabismic amblyopia, the excitatory contribution to binocular interactions is significantly reduced in both V1 and extra-striate cortex, while suppressive contributions remain intact. Our results provide robust electrophysiological evidence supporting the view that disruption of binocular interactions in strabismus or amblyopia is due to preferential loss of excitatory interactions between the eyes.
Journal
Year of Publication